PAI Life Sciences announced the dosing of the first healthy volunteer in a clinical trial of SchistoShield®, the company’s preventive vaccine against schistosomiasis, a major tropical disease threatening nearly 1 billion people in 79 countries with more than 260 million people currently infected.
Deadly tropical disease threatens hundreds of millions worldwide
SEATTLE--(BUSINESS WIRE)-- PAI Life Sciences, a biotechnology company specializing in translational research for neglected tropical diseases, today announced the dosing of the first healthy volunteer in a clinical trial of SchistoShield®, the company’s preventive vaccine against schistosomiasis, a major tropical disease threatening nearly 1 billion people in 79 countries with more than 260 million people currently infected.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20220531005051/en/
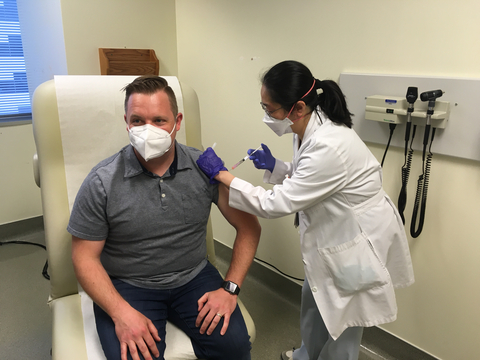
A study participant receives the first-ever shot of the SchistoShield® investigational vaccine at KPWHRI on May 23, 2022. © 2022 Kaiser Permanente Washington Health Research Institute
“Schistosomiasis is one of the deadliest tropical diseases in the world and is second only to malaria among parasitic diseases with the greatest devastating economic impact,” said PAI’s Vice President Sean Gray, Ph.D. “Developing an effective vaccine against schistosomiasis has been elusive, and while many vaccine candidates have been tested, few have offered any significant protection. We at PAI Life Sciences are humbled and excited at the prospect of our vaccine potentially making an impact in disease burden, as there is no effective vaccine to date.”
Schistosomiasis, also known as snail fever, is a disease caused by parasitic flatworms, called schistosomes. The worms enter human skin that comes in contact with contaminated fresh water. They are found in sub-Saharan Africa, Middle East, Southeast Asia and the Caribbean. Once in the bloodstream, the parasites usually first enter the urinary tract or the intestines. Initial symptoms include abdominal pain, bloody stools, diarrhea and blood in the urine. Long term infections can cause liver damage, kidney failure, infertility, bladder cancer and death.
The vaccine has shown to ameliorate disease, kills adult worms, and reduce fecundity in preclinical animal models, if proven clinically, could be a significant new tool added to the toolbox for global schistosomiasis elimination effort. The Phase 1, open-label, dose-escalation trial will evaluate the safety and the immunogenicity of PAI Life Sciences’s SchistoShield® vaccine candidate in 45 healthy adults between 18 and 55 years of age. Five treatment groups, each including nine subjects, will receive three intramuscular injections at different doses. One group will receive the vaccine without an adjuvant (which acts to increase immune responses) and four will receive the vaccine with an adjuvant. Estimated completion date is April 2024 (Visit ClinicalTrials.gov for additional details, Identifier: NCT05292391).
Professor Afzal A. Siddiqui, Ph.D., the lead researcher and inventor of this vaccine from the Texas Tech University Health Sciences Center and Dr. Darrick Carter, CEO of PAI Life Sciences Inc., said the SchistoShield® schistosomiasis vaccine has the potential to reduce the overall worm burden (prophylactic efficacy), lessen tissue pathology (anti-pathology efficacy) and decrease the infection transmission through reduction in output of mature eggs and diminished egg hatching (transmission-blocking efficacy). “An effective schistosomiasis vaccine potentially could benefit hundreds of millions of people in geographical areas ranging all across sub-Saharan Africa and parts of Asia and South America where schistosomiasis is endemic,” Siddiqui said. “Following Phase I trials in the U.S. and Phase 1b trials in Africa (Madagascar, Burkina Faso); vaccine efficacy trials in schistosome human challenge models in Europe (The Netherlands) and Africa (Uganda) will commence in 2023.”
The National Institute of Allergy and Infectious Diseases of the National Institutes of Health is sponsoring the Phase 1 trial. The study site is Kaiser Permanente Washington Health Research Institute, one of the NIAID- funded Vaccine and Treatment Evaluation Units; Lisa Jackson, MD, MPH, senior investigator, is the study principal investigator. PAI Life Sciences Inc. licensed the vaccine from the Texas Tech University Health Sciences Center.
About Texas Tech University Health Sciences Center (TTUHSC)
TTUHSC is a comprehensive institution of higher education that also provides patient care and conducts biomedical clinical research across six campuses in the state of Texas. The university, which is comprised of six schools (medicine, nursing, health professions, pharmacy, graduate school of biomedical sciences and population and public health), graduates more health care professionals than any other institution in the state. TTUHSC has been nationally recognized for innovative programs, academic achievement and innovation. The institution achieved Carnegie Classification® status for Special Focus Four-Year Research Institutions in 2022 and is a recognized Hispanic Serving Institution by the U.S. Department of Education. Founded in 1969, TTUHSC has trained more than 28,000 health care professionals to date and is a member of the Texas Tech University System. To learn more, visit www.ttuhsc.edu.
About PAI Life Sciences Inc.
PAI Life Sciences is a biotechnology company located in Seattle, WA, specialized in the developmental and translational research necessary to bring products from the laboratory to bedside. Its mission is to address neglected diseases throughout the world with the idea of “serving the underserved.” The company has developed novel protein biotherapeutics and has a pipeline of products ranging from infectious disease vaccines and adjuvants to therapeutics for cancer. For more information visit https://www.pailifesciences.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20220531005051/en/
Contacts
Darrick Carter, Ph.D.
darrick.carter@pailifesciences.com
Source: PAI Life Sciences Inc.
A study participant receives the first-ever shot of the SchistoShield® investigational vaccine at KPWHRI on May 23, 2022. © 2022 Kaiser Permanente Washington Health Research Institute
View this news release and multimedia online at:
http://www.businesswire.com/news/home/20220531005051/en