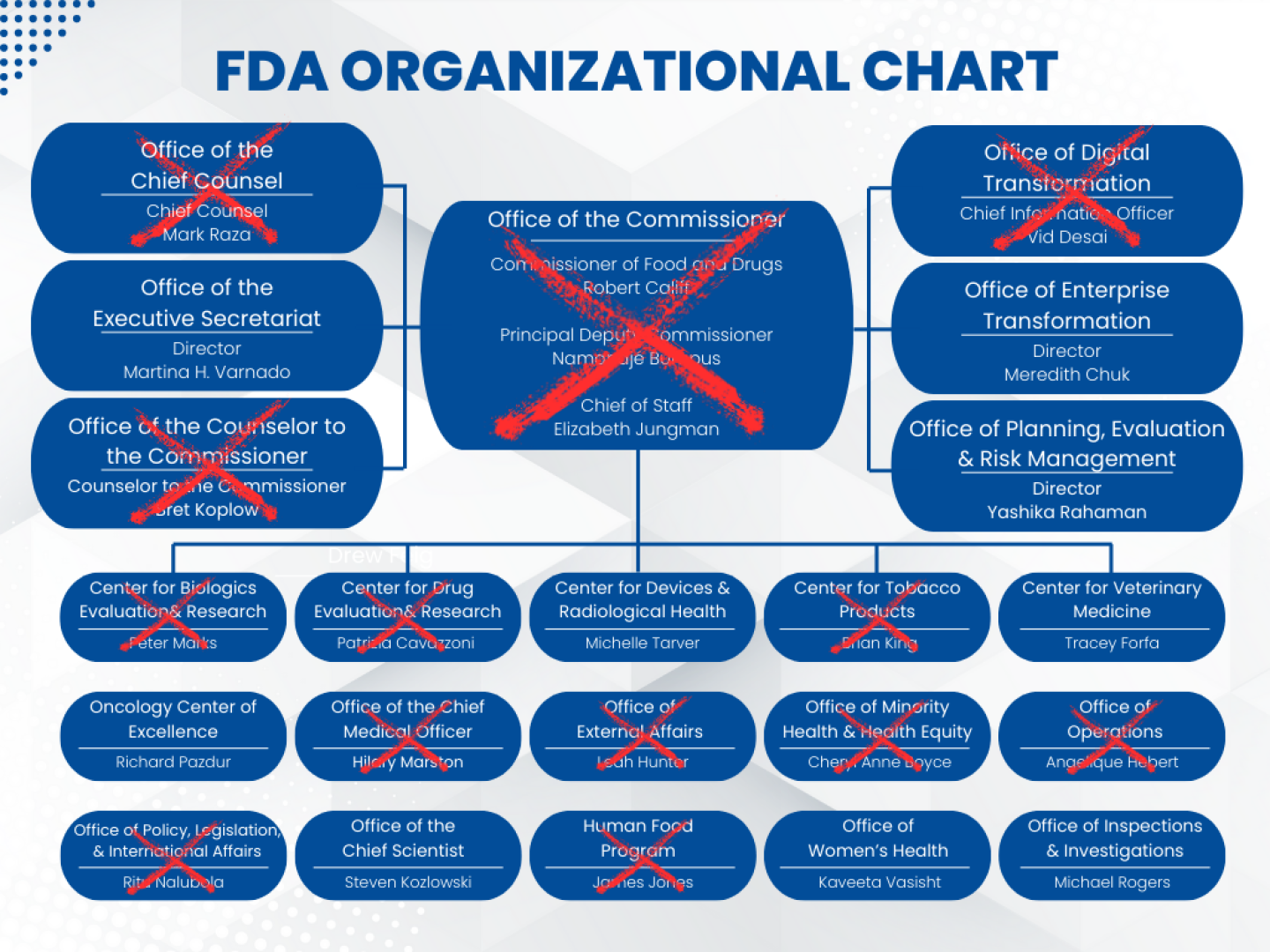
While it’s not unusual for certain positions to turn over with a new administration, the number of senior-level FDA staffers who have recently left the agency is unprecedented. The lack of communication, transparency and human decency is as well.
Former director of the FDA’s Center for Drug Evaluation and Research Patrizia Cavazzoni is looking pretty smart right now, having gotten out of the agency before what our colleague Annalee Armstrong has dubbed “the Marks massacre.”
Cavazzoni—who resigned as head of CDER two days before Donald Trump was sworn in again as U.S. president—wasn’t alone in abandoning the soon-to-be sinking ship. She followed Namandjé Bumpus, the agency’s principal deputy commissioner, out the door, and they were joined by Douglas Throckmorton, deputy director for regulatory programs, and Robert Temple, CDER’s senior advisor for clinical science, both of whom retired prior to Robert F. Kennedy Jr. taking the helm at the Department of Health and Human Services.
These were prescient moves by individuals who may have seen the writing on the proverbial wall.
Of all the cuts levied at the FDA, none have rocked the world of biopharma more than the departure of Peter Marks, director of the Center for Biologics Evaluation and Research and a well-respected regulator across the industry. When that news broke late last Friday, a day after Kennedy announced 10,000 job cuts to come at HHS, diverse voices expressed disbelief, fear and outrage. Former FDA Commissioner Robert Califf put it bluntly in a LinkedIn message: “The FDA as we’ve known it is finished.”
Unfortunately, Marks is not the only senior leader at the FDA to get the boot. More than half of the FDA’s senior leadership has exited the agency in the past six months, and STAT News counts two dozen employees of director level or above who have recently departed the agency.
“Most of the leaders with institutional knowledge and a deep understanding of product development and safety [are] no longer employed,” lamented Califf in his LinkedIn message Tuesday. “I believe that history will see this a huge mistake.”
Working down the ladder, the cuts only get more numerous. It’s perfectly normal for a new administration to come in and make changes. FDA commissioner, for example, is an appointed position that often turns over with the White House. Califf stepped down on Inauguration Day in January, and Marty Makary was soon nominated in his stead and confirmed last Wednesday.
But other staffing changes at the FDA are decidedly not normal. Just two days after her appointment was announced earlier this month, Chief Counsel Hilary Perkins announced on X that she was leaving, Reuters reported, and several other senior-level employees have recently left the agency, including Chief Medical Officer Hilary Marston and Peter Stein, director of CDER’s Office of New Drugs.
Democratic leaders are now pushing back, with Senator Cory Booker (D-NJ) delivering a record-breaking 25-hour floor speech through the night on Tuesday and into Wednesday evening. Analysts at the firm Cantor Fitzgerald this week went so far as to call for the removal of RFK Jr. as HHS Secretary. A poll BioSpace posted to LinkedIn shows that many support this action.
Marks had expressed his intention to stay in his role under the new Trump administration despite holding views that conflicted with Kennedy’s, but in the end, he was forced out against his will. And the planned HHS layoffs did not initially include CDER or OND leadership, Peter Stein, recently ousted director of the CDER’s Office of New Drugs, told BioPharma Dive.
As for what the future holds for the FDA, it’s anyone’s guess, but interested parties are dreading worst-case scenarios. In a note to investors on Wednesday, Leerink analysts wrote, “we fear FDA will bring more conflicting voices to bear who challenge proven science.” For now, the markets have not responded positively. When Marks got the boot—something analysts called “arguably biotech investors’ greatest fear”—the S&P Biotech Index, or XBI, slid to its lowest point since December 2023.
Those left at the FDA are also living in fear. “It’s tense here now despite receiving word of mouth that the cuts are complete,” according to one source within the agency. “Most people keep their doors closed.”
Thousands Gone From FDA
While this week’s massive HHS layoffs garnered the most attention, they weren’t the first of this administration. FDA staffers have been on tenterhooks since mid-February, when an unknown number of employees, primarily in the agency’s centers for food, medical devices and tobacco products, were axed days after Kennedy was confirmed as HHS Secretary. Employees at the National Institutes of Health (NIH) and Centers for Disease Control and Prevention (CDC) were also let go.
There were inklings of discontent from biopharma leaders even then, with many calling for transparency. “The extent and the rapidity of the actions taken has been remarkable, and that has perhaps led to, in many cases, the concern that there is no understanding of exactly why they’re being done,” Ovid Therapeutics CEO Jeremy Levin told BioSpace at the time.
It seems these calls are not being heeded—either within the agency or to the public. Many FDA employees learned of their terminations only after showing up to work on Tuesday and discovering that their badges wouldn’t work.
“[T]here is no good reason to treat people this way,” Califf wrote on LinkedIn Tuesday.
In his welcome reception speech to much of the agency on Wednesday, Makary said that letting go of nearly 20% of FDA staff was a hard but critical step of RFK Jr.’s “Making America Healthy Again” campaign, Endpoints reported. He also talked about the importance of being transparent with the public.
That will likely prove difficult as the FDA’s communications teams have been dismantled. According to Erica Jefferson, senior vice president of corporate affairs at ReCode Therapeutics, the main FDA media team as well as those at CBER, CDER, and other divisions of the agency are all but gone. Groups that handle congressional communications and website publications are also gone, as are many FOIA staff.
Indeed, an email we sent to an agency communications representative came back with an automatic response that they were “on administrative leave,” and there has only been one press release put out by the FDA this week: on April 1, about Makary’s welcome ceremony on Wednesday—four days after he was apparently secretly sworn in and gave the green light to can Marks.
As for the ceremony itself, FDA staffers reportedly began setting up for it on Monday. “It’s hard to wrap my head around the sequence,” an FDA insider said: “set up for the commissioner Monday evening, allow personnel to begin commuting on April Fools day while their RIF notices reached their unmonitored inboxes, then about face and pretend that Wednesday is a day of celebration.”
The Skeleton Crew
The fallout of the Marks massacre remains to be seen. Immediately following Marks’ resignation, Leerink analysts wondered, “Will new FDA Commissioner Marty Makary . . . be able to support scientifically-proven research and products and effectively manage upward with RFK Jr., or will he ultimately face the same fate as Dr. Marks?”
Former FDA leaders including Scott Gottlieb and Mark McClellan told BioCentury’s Steve Usdin that the layoffs could hamstring the agency’s functioning, according to a LinkedIn post by Usdin’s colleague Jeff Cranmer. This includes interfering with “the FDA’s ability to make determinations that new and generic drugs are safe and effective,” Cranmer summarized.
Sure enough, the FDA has already missed a PDUFA deadline for the latest formulation of Novavax’s COVID-19 shot after Sara Brenner, the FDA’s recently appointed principal deputy commissioner, reportedly intervened.
Another function likely to be impeded is the FDA’s inspections of manufacturing facilities, including those overseas, despite assurances from Kennedy during his confirmation hearing that the inspectors would not be touched. According to reporting from Vanity Fair, the FDA has asked some laid-off employees to temporarily return to work to allow such inspections to be carried out. On Thursday, Kennedy admitted that 20% of the most recent job cuts could be wrong.
“Personnel that should not have been cut, were cut. We’re reinstating them. And that was always the plan,” Kennedy said, according to CBS News.
We’ll see if these reinstatements play out, but if we have any hope, it’s with the people who remain at FDA. This includes Oncology Center of Excellence Director Richard Pazdur, who told Endpoints News this week, “I have no plans on leaving.” Of course, neither did Marks. Pazdur, and others left behind, undoubtedly face a challenging road ahead.
As we watch the chaos unfold, our hearts go out to all the federal employees caught up in the drama, both those who’ve been cut unceremoniously and those left behind to pick up the pieces without support and without transparency about the reasoning for the upheaval. The industry is behind them as well.
ReCode CEO Shehnaaz Suliman captured this sentiment on LinkedIn this week when she wrote, “I, like most, stand in support of those who remain who believe in Peter’s vision and hopefully will be allowed to continue to advance the FDA’s work.”