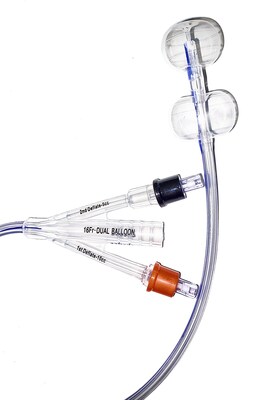
HR Pharmaceuticals, Inc. announced that they have entered into an Exclusive Commercial Agreement with Poiesis Medical LLC, under which HRP will license Poiesis’s Dual Balloon Catheter.
YORK, Pa., Dec. 28, 2023 /PRNewswire/ -- Today, HR Pharmaceuticals, Inc. (HRP) announced that they have entered into an Exclusive Commercial Agreement with Poiesis Medical LLC, under which HRP will license Poiesis's Dual Balloon Catheter (Duette™). Under the terms of the agreement, HRP will have exclusive commercialization rights in North America. This product expands HRP's growing urological portfolio across the continuum of care, providing a novel design to help address adverse events associated with Foley catheterization, which is critically important for specific patient populations.
"The Dual Balloon Catheter has demonstrated significant benefits compared to traditional Foley catheters, leading to better patient outcomes. Specifically, we see a great opportunity for patients requiring indwelling catheterization greater than five days. This product will become an important part of our broader urology strategy over the next 12 months," states Colby Wiesman, President of HR Pharmaceuticals, Inc.
"The Dual Balloon Catheter technology, a brainchild of my father, Dr. Bruce Wiita, a practicing urologist and decorated Vietnam veteran, was born out of a genuine desire to improve the quality of life for patients enduring prolonged catheterization and to alleviate the strains it places on the healthcare system," says Gregory Wiita, CEO and Founder at Poiesis Medical LLC. "We are excited about this new chapter with HR Pharmaceuticals and the potential of the technology to make a meaningful impact in patient care."
HRP will begin processing North American orders for the Duette Dual Balloon Catheters starting January 2, 2024. Duette is available at most national distributors, including Cardinal Health, McKesson, Medline, and Owens & Minor. For further assistance or additional information, please contact HRP at customercare@hrpharma.com or 800.302.1110.
About HR Pharmaceuticals, Inc.
HR Pharmaceuticals, Inc. is a professionally managed, family-owned business located in York, PA, that develops, manufactures, and supplies products for a variety of markets. HR is an industry expert in healthcare consumables and offers a broad range of products, from medical lubricants and ultrasound gels to a comprehensive continence care offering and more. HR's mission - positively impacting people's lives- unifies the organization and drives an unwavering commitment to customers. For more information on HR Pharmaceutical, Inc., visit hrpharma.com.
About Poiesis Medical LLC
Poiesis Medical LLC is a family-oriented business co-founded by Dr. Bruce Wiita and his son Gregory Wiita. The father-son team has been at the forefront of medical innovation for 25 years. The company's journey spans several successful ventures, marked by significant contributions to robotic radiotherapy in prostate cancer care and developing safety devices for laparoscopic lymphadenectomy surgery. With a history of successful partnerships and exits, including three acquisitions, the legacy of innovation continues with a focus on delivering advanced urinary products to provide better patient care.
Duette is a trademark of Poiesis Medical LLC.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/hr-pharmaceuticals-inc-receives-exclusive-commercialization-rights-to-poiesis-medicals-dual-balloon-catheter-technology-in-north-america-302022947.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/hr-pharmaceuticals-inc-receives-exclusive-commercialization-rights-to-poiesis-medicals-dual-balloon-catheter-technology-in-north-america-302022947.html
SOURCE HR Pharmaceuticals