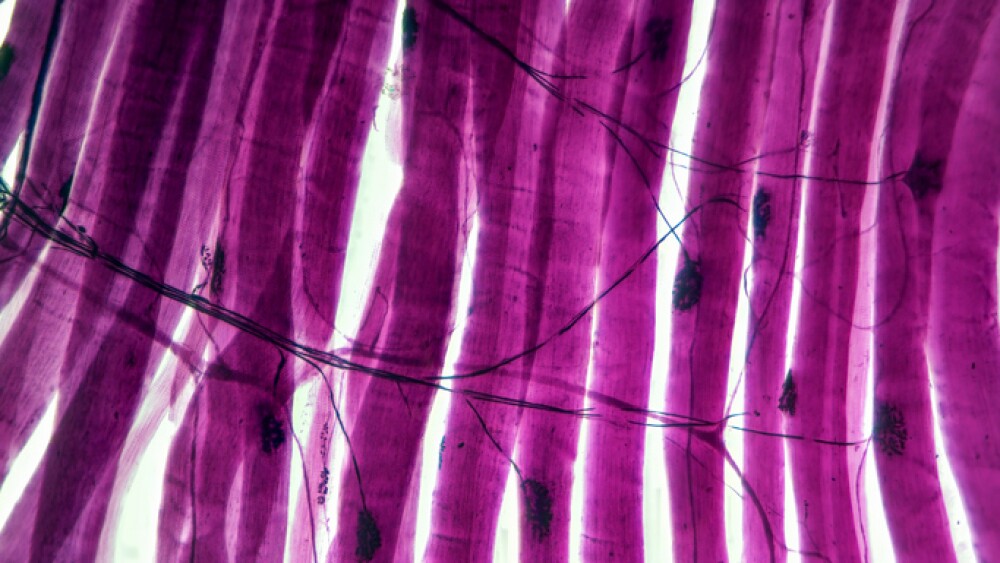
Presenting new clinical data from Phio’s on-going Phase 1b trial
Marlborough, Massachusetts--(Newsfile Corp. - November 7, 2024) - Phio Pharmaceuticals Corp. (NASDAQ: PHIO) is a clinical-stage biotechnology company creating new pathways towards a cancer-free future by using its INTASYL® siRNA gene silencing technology designed to make the body's immune cells more effective in killing cancer cells. Phio announced today that it is presenting clinical data from its on-going Phase 1b clinical trial at the 39th Annual Meeting of the Society for Immunotherapy of Cancer (SITC), which will be held in Houston, TX from November 8-10, 2024.
Phio's presentation will highlight the clinical results from enrolled patients in Cohorts 1 and 2 in the Phase 1b dose escalation clinical trial. This clinical trial is designed to evaluate the safety and tolerability of intratumoral injection of PH-762 in patients with cutaneous squamous carcinoma, Merkel cell carcinoma and melanoma.
Clinical Results To Date
Intratumoral (IT) injection of PH-762 has been well tolerated in all patients enrolled in the trial to date. There were no related adverse events, no serious adverse events, and no dose limiting toxicities or dose adjustments. Five (5) patients with cutaneous carcinomas have enrolled in Cohorts 1 and 2. Each enrolled patient received four (4) IT doses of PH-762. The patients were diagnosed with cutaneous squamous cell carcinoma (4 patients) and metastatic melanoma (1 patient).
At Day 36 (tumor excision), while each patient in the first cohort had stable disease, the following results were described for two patients enrolled in the second cohort:
"We are encouraged by the documented clinical and histopathological results observed to date," said Mary Spellman MD, Phio's acting Chief Medical Officer. "Corresponding safety and tolerability data supports continued assessment of intratumoral PH-762 in patients with cutaneous carcinomas."
Presentation Details are as follows:
About Phio Pharmaceuticals Corp.
Phio Pharmaceuticals Corp. (NASDAQ: PHIO) is a clinical-stage biotechnology company and a pioneer in the RNAi revolution advancing its proprietary INTASYL siRNA gene silencing technology to create new pathways towards a cancer-free future. INTASYL can target and silence virtually any gene with a high degree of specificity across a wide range of cell types and tissues. INTASYL is designed to enhance the ability of immune cells to more effectively kill tumor cells. INTASYL has also been used to enhance adoptive cell therapy. Notably, INTASYL is a self-delivering RNAi technology focused on immuno-oncology therapeutics without the need for formulation enhancements or manipulations to reach its target.
Phio's lead clinical program, PH-762, is an INTASYL compound that silences PD-1. PH-762 is a potential non-surgical treatment for skin cancers. The on-going Phase 1b trial (NCT# 06014086) received FDA clearance for an Investigational New Drug Application to evaluate PH-762 in the treatment of cutaneous SCC, melanoma and Merkel cell carcinoma in the second quarter of 2023.
For additional information, visit the Company's website, www.phiopharma.com.
Forward Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements can be identified by words such as "intends," "believes," "anticipates," "indicates," "plans," "expects," "suggests," "may," "would," "should," "potential," "designed to," "will," "ongoing," "estimate," "forecast," "target," "predict," "could" and similar references, although not all forward-looking statements contain these words. Examples of forward-looking statements contained in this press release include, among others, the possibility that our INTASYL® siRNA gene silencing technology will make the body’s immune cells more effective in killing cancer cells and statements regarding our commercial and clinical strategy, development plans and timelines and other future events.
These statements are based only on our current beliefs, expectations and assumptions and are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict and many of which are outside of our control. Our actual results may differ materially from those indicated in the forward-looking statements as a result of a number of important factors, including, but not limited to, the impact to our business and operations by inflationary pressures, rising interest rates, recession fears, the development of our product candidates, results from our preclinical and clinical activities, our ability to execute on business strategies, our ability to develop our product candidates with collaboration partners, and the success of any such collaborations, the timeline and duration for advancing our product candidates into clinical development, the timing or likelihood of regulatory filings and approvals, the success of our efforts to commercialize our product candidates if approved, our ability to manufacture and supply our product candidates for clinical activities, and for commercial use if approved, the scope of protection we are able to establish and maintain for intellectual property rights covering our technology platform, our ability to obtain future financing, market and other conditions and those identified in our Annual Report on Form 10-K and subsequent Quarterly Reports on Form 10-Q under the caption "Risk Factors" and in other filings the Company periodically makes with the SEC. Readers are urged to review these risk factors and to not act in reliance on any forward-looking statements, as actual results may differ from those contemplated by our forward-looking statements. Phio does not undertake to update forward-looking statements to reflect a change in its views, events or circumstances that occur after the date of this release, except as required by law.
Contact:
Phio Pharmaceuticals Corp.
Jennifer Phillips: jphillips@phiopharma.com
Corporate Affairs
To view the source version of this press release, please visit https://www.newsfilecorp.com/release/228924
Phio's presentation will highlight the clinical results from enrolled patients in Cohorts 1 and 2 in the Phase 1b dose escalation clinical trial. This clinical trial is designed to evaluate the safety and tolerability of intratumoral injection of PH-762 in patients with cutaneous squamous carcinoma, Merkel cell carcinoma and melanoma.
Clinical Results To Date
Intratumoral (IT) injection of PH-762 has been well tolerated in all patients enrolled in the trial to date. There were no related adverse events, no serious adverse events, and no dose limiting toxicities or dose adjustments. Five (5) patients with cutaneous carcinomas have enrolled in Cohorts 1 and 2. Each enrolled patient received four (4) IT doses of PH-762. The patients were diagnosed with cutaneous squamous cell carcinoma (4 patients) and metastatic melanoma (1 patient).
At Day 36 (tumor excision), while each patient in the first cohort had stable disease, the following results were described for two patients enrolled in the second cohort:
- Complete response (100% tumor clearance) reported for 1 patient with cutaneous squamous cell carcinoma; and
- Partial response (90% tumor clearance) reported for 1 patient with cutaneous squamous cell carcinoma.
"We are encouraged by the documented clinical and histopathological results observed to date," said Mary Spellman MD, Phio's acting Chief Medical Officer. "Corresponding safety and tolerability data supports continued assessment of intratumoral PH-762 in patients with cutaneous carcinomas."
Presentation Details are as follows:
| Title: | INTASYL PH-762: PD-1 Directed Intratumoral Immunotherapy for Cutaneous Carcinoma |
| Abstract Number: | 696 |
| Presenting Author: | Mary Spellman, M.D. |
| Date: | Saturday November 9, 2024 |
| Location: | Exhibit Halls A and B, George R. Brown Convention Center |
About Phio Pharmaceuticals Corp.
Phio Pharmaceuticals Corp. (NASDAQ: PHIO) is a clinical-stage biotechnology company and a pioneer in the RNAi revolution advancing its proprietary INTASYL siRNA gene silencing technology to create new pathways towards a cancer-free future. INTASYL can target and silence virtually any gene with a high degree of specificity across a wide range of cell types and tissues. INTASYL is designed to enhance the ability of immune cells to more effectively kill tumor cells. INTASYL has also been used to enhance adoptive cell therapy. Notably, INTASYL is a self-delivering RNAi technology focused on immuno-oncology therapeutics without the need for formulation enhancements or manipulations to reach its target.
Phio's lead clinical program, PH-762, is an INTASYL compound that silences PD-1. PH-762 is a potential non-surgical treatment for skin cancers. The on-going Phase 1b trial (NCT# 06014086) received FDA clearance for an Investigational New Drug Application to evaluate PH-762 in the treatment of cutaneous SCC, melanoma and Merkel cell carcinoma in the second quarter of 2023.
For additional information, visit the Company's website, www.phiopharma.com.
Forward Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements can be identified by words such as "intends," "believes," "anticipates," "indicates," "plans," "expects," "suggests," "may," "would," "should," "potential," "designed to," "will," "ongoing," "estimate," "forecast," "target," "predict," "could" and similar references, although not all forward-looking statements contain these words. Examples of forward-looking statements contained in this press release include, among others, the possibility that our INTASYL® siRNA gene silencing technology will make the body’s immune cells more effective in killing cancer cells and statements regarding our commercial and clinical strategy, development plans and timelines and other future events.
These statements are based only on our current beliefs, expectations and assumptions and are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict and many of which are outside of our control. Our actual results may differ materially from those indicated in the forward-looking statements as a result of a number of important factors, including, but not limited to, the impact to our business and operations by inflationary pressures, rising interest rates, recession fears, the development of our product candidates, results from our preclinical and clinical activities, our ability to execute on business strategies, our ability to develop our product candidates with collaboration partners, and the success of any such collaborations, the timeline and duration for advancing our product candidates into clinical development, the timing or likelihood of regulatory filings and approvals, the success of our efforts to commercialize our product candidates if approved, our ability to manufacture and supply our product candidates for clinical activities, and for commercial use if approved, the scope of protection we are able to establish and maintain for intellectual property rights covering our technology platform, our ability to obtain future financing, market and other conditions and those identified in our Annual Report on Form 10-K and subsequent Quarterly Reports on Form 10-Q under the caption "Risk Factors" and in other filings the Company periodically makes with the SEC. Readers are urged to review these risk factors and to not act in reliance on any forward-looking statements, as actual results may differ from those contemplated by our forward-looking statements. Phio does not undertake to update forward-looking statements to reflect a change in its views, events or circumstances that occur after the date of this release, except as required by law.
Contact:
Phio Pharmaceuticals Corp.
Jennifer Phillips: jphillips@phiopharma.com
Corporate Affairs
To view the source version of this press release, please visit https://www.newsfilecorp.com/release/228924