SCYNEXIS, Inc. is conducting a voluntary nationwide recall of 2 lots of BREXAFEMME® to the consumer level in the US market due to potential cross contamination with a non-antibacterial ß-lactam drug substance in the ibrexafungerp citrate used to manufacture the BREXAFEMME® tablets.
JERSEY CITY, N.J., Sept. 27, 2023 (GLOBE NEWSWIRE) -- SCYNEXIS, Inc. is conducting a voluntary nationwide recall of 2 lots of BREXAFEMME® (ibrexafungerp tablets) to the consumer level in the US market due to potential cross contamination with a non-antibacterial ß-lactam drug substance in the ibrexafungerp citrate used to manufacture the BREXAFEMME® tablets. During a review of manufacturing equipment and cleaning activities at a supplier, SCYNEXIS was made aware of potential cross-contamination risk with a non-antibacterial beta-lactam drug substance. This press release provides additional details on the voluntary product recall recently disclosed by SCYNEXIS.
Risk Statement: The potential cross contamination with a non-antibacterial beta-lactam drug substance could lead to hypersensitivity reactions such as swelling, rash, urticaria and anaphylaxis, a potentially life-threatening adverse reaction. To date, SCYNEXIS has not received any reports of adverse events established to be due to the possible beta-lactam cross contamination.
BREXAFEMME® is an antifungal product indicated for the treatment of vulvovaginal candidiasis (VVC) and the reduction of the incidence of recurrent vulvovaginal candidiasis (RVVC).
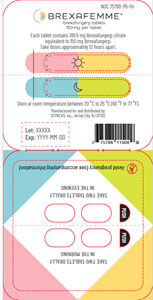
BREXAFEMME® is dispensed in a carton and packaged in blister packs with four 150-mg tablets (NDC 75788-115-04). BREXAFEMME® tablet for oral administration is a purple, oval, biconvex shaped, film-coated tablet debossed with 150 on one side and SCY on the other side. The affected BREXAFEMME® lots include the following lots and expiration dates: LF21000008 (expiration date 11/2023) and LF22000051 (expiration date 11/2025). The recalled lots were distributed nationwide to wholesalers across the US, beginning in December 2022.
SCYNEXIS is engaging with Sedgwick to manage the recall of the product down to the consumer level. Sedgwick will be notifying BREXAFEMME® distributors via a recall notification letter and will be arranging for the return of the recalled lot from distributors, retailers, and consumers.
Consumers with questions regarding this recall can contact Sedgwick at 1-877-551-7154. Office hours: Monday to Friday, 8:00 AM to 5:00 PM ET.
Consumers should contact their healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Contact Information:
Consumer:
Sedgwick
1-877-551-7154
Media:
media@scynexis.com
Reference
BREXAFEMME (prescribing information), Jersey City, NJ: SCYNEXIS, Inc., 2022
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/3a914236-b706-460c-bb31-2162cbf4ff16
https://www.globenewswire.com/NewsRoom/AttachmentNg/c456f9be-c79c-4e4e-9807-bcb1ea4f91ba